Genetic Risk
T1D starts with genetic risk– genes that put you at higher risk for the disease
T1D starts with a genetic predisposition, meaning you have certain genes that put you at higher risk. Everyone who develops T1D has these genes, including people with no family history of T1D. The risk for people in the general population (no T1D family history) is about 1 in 300. For those who have a family member with T1D, the risk is 1 in 20, or 15x greater.

Know Your Risk
TrialNet Risk Screening
Available at no cost to relatives of people with T1D, TrialNet risk screening identifies T1D in its earliest stages, often years before symptoms appear. Early detection allows you to take steps to try to change the course of the disease. TrialNet offers prevention studies testing ways to slow or stop disease progression. For people who take part in clinical trials, the risk of diabetic ketoacidosis (DKA)—a serious and potentially life-threatening condition—drops from 30% to less than 3%. Learn more


Autoantibodies
T1D autoantibodies are proteins that mark insulin producing beta cells for destruction. They are a useful tool for identifying people at risk of developing T1D, as they are easily identified in the blood. TrialNet can test for up to five T1D autoantibodies.
Immune Activation/Response
The role of autoantibodies in the development of T1D
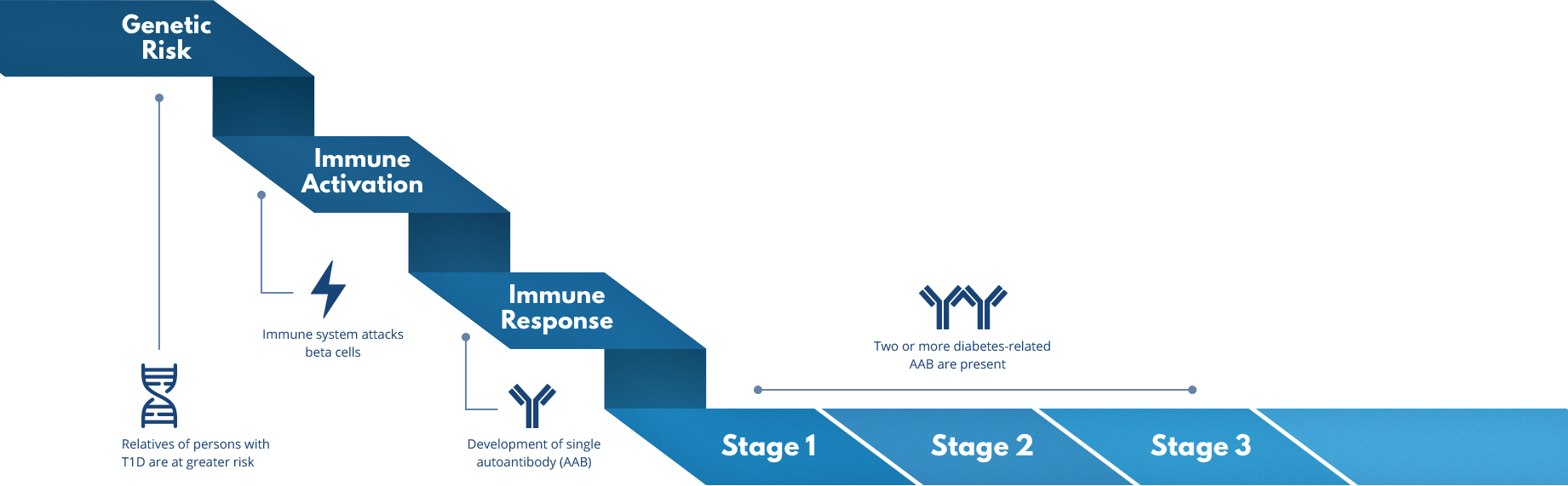
During the immune activation and immune response involved in T1D, the immune system mistakenly attacks cells in the pancreas (immune activation), leading to the development of a single diabetes-related autoantibody (immune response). Almost all individuals who develop two or more of these diabetes-related autoantibodies (stage 1 and 2) will eventually be diagnosed with T1D (stage 3). But TrialNet is working to change that.
RESEARCH OPPORTUNITIES
TrialNet Prevention Studies
If your screening results show you are in the early stages of T1D, you may be able to participate in a TrialNet prevention study. Prevention studies are testing ways to slow or stop disease progression by preserving insulin production. Learn more


Beta Cells
Located in the pancreas, beta cells produce insulin. Beta cells are the target of the auto-immune response in T1D.
Stage 1
T1D progresses in three stages. Stage 1 is the start.
In stage 1, individuals test positive for two or more diabetes-related autoantibodies, identified by TrialNet T1D risk screening. The immune system has started attacking insulin-making beta cells. Blood sugar levels remain normal and no symptoms are present.


Rate of Progression
Research shows that age plays a key role in how fast T1D progresses. Younger children typically progress faster through the stages of T1D than adults. TrialNet researchers are studying additional markers to more accurately predict who will develop T1D and when.
Stage 2
Blood sugar levels become abnormal in stage 2
In stage 2, individuals have two or more diabetes-related autoantibodies, but now blood sugar levels have become abnormal. This is due to an increased loss of beta cells. Still, there are often no symptoms.


Diabetic Ketoacidosis
Diabetic ketoacidosis (DKA), is a serious and potentially life-threatening condition that occurs when the body can’t make enough insulin and blood sugar becomes dangerously high. For people taking part in T1D research, the risk of DKA at diagnosis drops from 30% to less than 3%.
Stage 3
Stage 3 used to be known as the start of T1D. Now we know it is clinical diagnosis.
In stage 3, T1D symptoms are present due to significant beta cell loss. Common symptoms include increased thirst and urination, unexplained weight loss, blurred vision, and fatigue. Some people are diagnosed with T1D when they are hospitalized with diabetic ketoacidosis (DKA), a serious and potentially life-threatening condition.
RESEARCH OPPORTUNITIES
Studies for Newly Diagnosed, Stage 3 T1D
If you or a family member is newly diagnosed with T1D (stage 3), you may be eligible for a clinical study testing therapies to preserve insulin production. Learn more